Thermochemistry
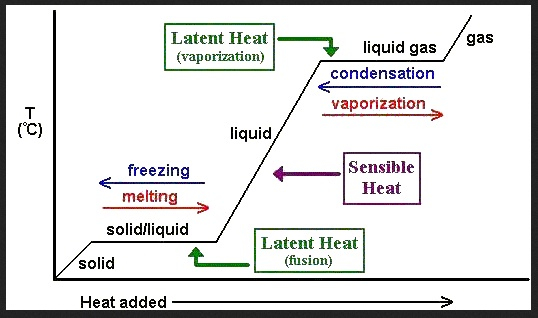
Thermochemistry has been defined as “the branch of chemistry concerned with the study and measurement of the heat evolved or absorbed during chemical reactions,” ("Thermochemistry", par 1). This can be quantitatively assessed through the measurement of the enthalpies of fusion, vaporization and atomization. These values are measures in kilojoules per mole (kJ/mol). For Neodymium, these values are 7.1 kJ/mol, 285 kJ/mol, and 328 kJ/mol respectively. These are relatively low as the enthalpies for Iron are 13.8 kJ/mol, 347 kJ/mol, and 415 kJ/mol.