Periodicity
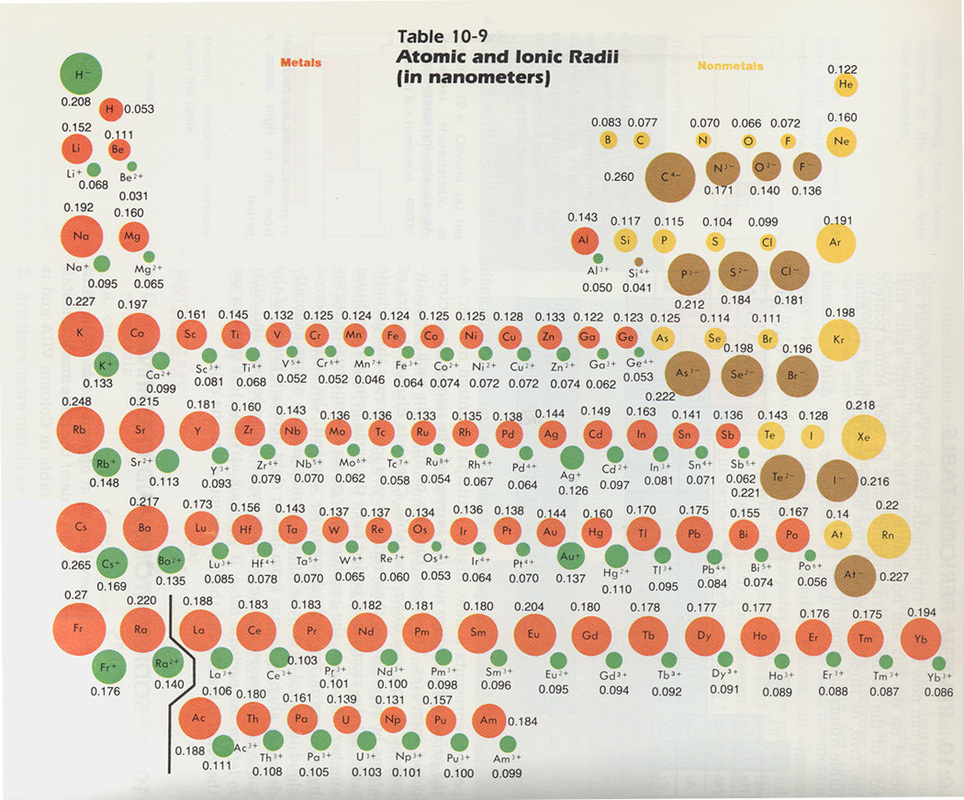
Periodicity of an element on the periodic table refers mostly to the periodic trends that can be seen in ionization energy, atomic radius, electronegativity, and electron affinity. The ionization energy is the "amount of energy required to remove an electron from an atom." The atomic radius of an atom is the “half the distance between the center of two atoms of that are touching each other” Electronegativity is the “measure of the ability of an atom to form chemical bonds.” Finally, electron affinity is the “ability of an atom to accept an electron.” Neodymium’s ionization number, electronegativity, electron affinity and atomic radius are 533.1 kilojoules (kJ) per mole, 1.14, 50 kJ per mole, and 143 pm for Neodymium 2+ atoms and 112.3 pm for Neodymium 3+ atoms respectively.