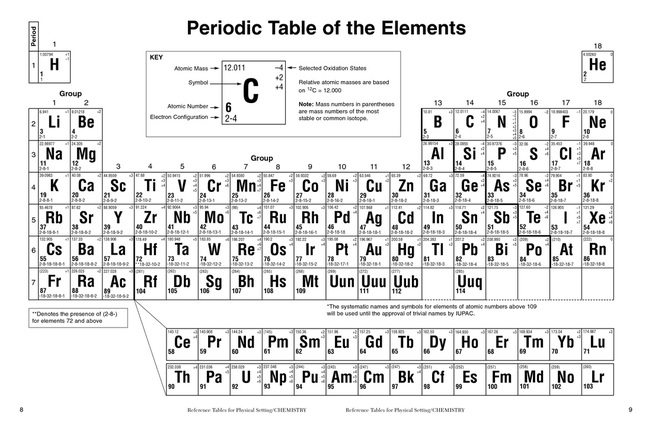
Oxidation States
The Oxidation state of an element is the charge that it takes in a compound with other elements and ions. For example, a regular sulfur atom may have a charge of 2- naturally on the periodic table because of its placement in the 16th group. However, when it is bonded to four oxygen atoms that always have the oxidation state of 2- to form sulfate, sulfur must have a 6+ charge to form the 2- charge that the sulfate ion (SO₄) has. Similarly, Neodymium can have the oxidation states of 2+ to 3+ depending on the element or ion it is bonded to. For the most part though, Neodymium binds at the oxidation state of 3+.