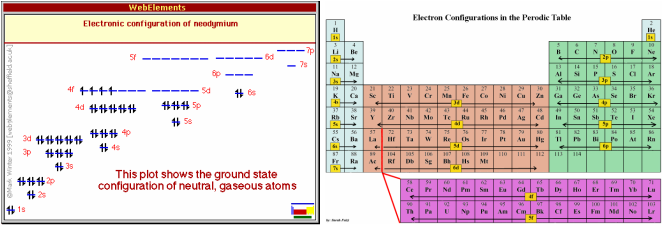
Electron Configuration
An element’s electron configuration is the very important when understanding the properties of the element. Each element is assigned a the same number of electrons as its atomic number or its number of protons originally. However, as this element’s atoms are reacted with other atoms and molecules, these electrons may be gained, lost or shared with other atoms. An element’s electron count can be expressed in many different ways, especially orbital notation, electron configuration and noble gas notation. Neodymium has 60 electrons and its electron configuration is shown below.